Join our email list to stay up to date on the latest Amyloidosis news.
Johnny Rosa
Marketing & Communications Manager
Amyloidosis Research Consortium
May is Mental Health Awareness Month. For those living with amyloidosis, the psychological toll of a rare diagnosis is intense, yet often goes unnamed. The following story is about surviving a disease for two decades, as well as a map of spiritual and mental evolution.
 Suri Harish meditating with his wife.
Suri Harish meditating with his wife.
The night before his world changed, everything seemed fine. It was 2006, and Suri Harish was celebrating his son’s birthday. There was lasagna, red wine, and the warmth of family, but later that night the celebration ended in illness. When his wife saw him throw up blood, she knew something was wrong, but Suri brushed it off. “It’s just the wine,” he insisted. “What else could it be?”
Due to persistent back pain, Suri visited his chiropractor in the morning, but his wife insisted on a visit to his primary care physician. Within days, a biopsy confirmed AL (Light Chain) amyloidosis. Within weeks, a specialist delivered a second blow: Suri also had multiple myeloma, later diagnosed as AL Amyloidosis with associated multiple myeloma.
He was told he had two to two and a half years to live, based on his diagnosis by a specialist.
“My wife and I walked to our car sobbing,” Suri recalls. He was 60 years old, an active man who didn’t even take aspirin. His parents had lived into their 90s. The diagnosis felt like an impossibility that could never have happened to him.
Looking back, Suri frames the two decades since his diagnosis in four distinct emotional phases. They didn’t arrive on a schedule, but they all deeply impacted him.
Phase One: “Why Me?”
The physical diagnosis was swift, and the initial mental impact was a “treacherous time of desperate and painful emotions.” Suri calls this period Phase One.
“It was scary,” Suri says. “The biggest thing in my mind was, ‘Why me?’ I’m a healthy guy. I exercise. I did everything right.”
He joined a support group, seeking community, but found that it often deepened his despair. As fellow members passed away, Suri felt he was simply catching a glimpse of his own inevitable end. When he was referred to the Mayo Clinic in January 2007, he and his wife rented the best furnished apartment they could find, physically connected to Mayo Clinic, regardless of the $4,800 monthly price tag.
“What’s the money for?” he thought. “I’m going to die anyway.” At the time, the psychology team evaluated him and concluded he wasn’t emotionally ready for a stem cell transplant. Suri agreed. He was trapped in the gravity of Phase One.
Phase Two: The Will to Fight
The turning point arrived on the day of his transplant. Suri looked around his hospital room and saw his doctors and nurses, his wife, sons, and sister, as well as a priest named Mary (who had cleared her schedule to be there) and a second chaplain Mary had sent in case she ran late. All 10 of these people were there to support him and raise his spirits.
“It hit me,” Suri says. “This wouldn’t be happening if God didn’t want me to fight. This was my clue.”
The “why me?” transformed into “fight mode” and Phase Two began. Over the next year, Suri found that constant fighting was exhausting for the winner and the loser alike. Even as his body improved, he felt emotionally drained. He began to ask himself, “This disease is a part of me. If I am fighting it, am I fighting myself?”
Slowly, the anger began to soften into self-compassion.
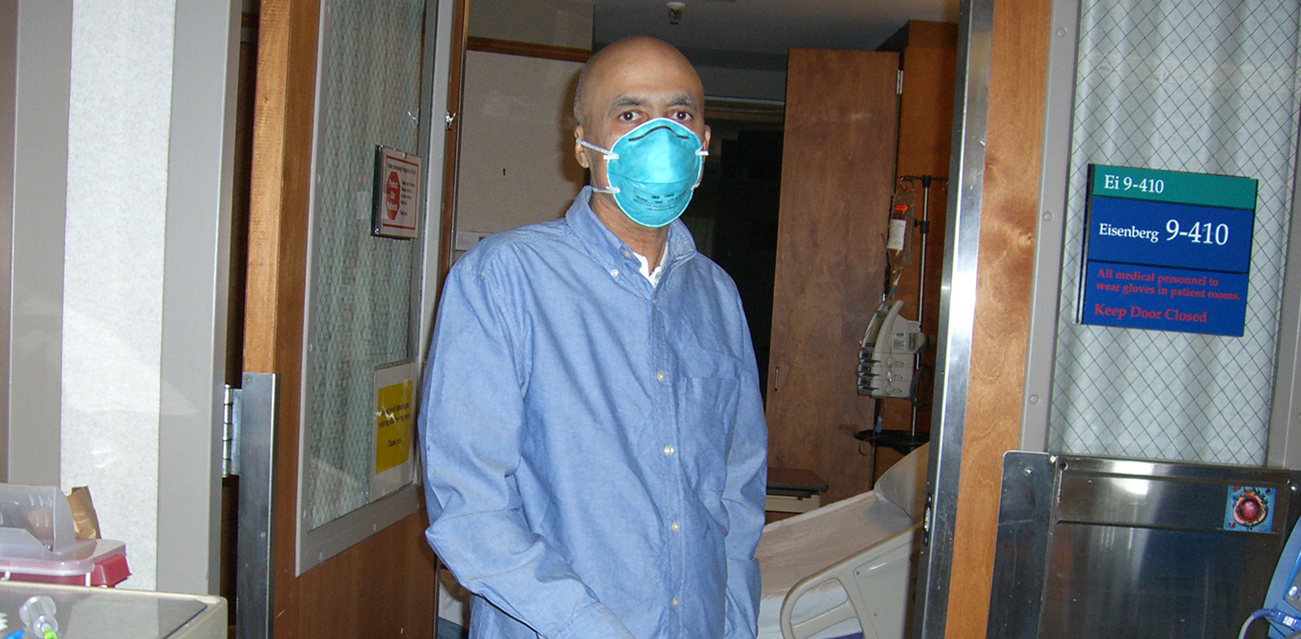 Suri fighting at the hospital.
Suri fighting at the hospital.
Phase Three: From Fighting to Living
Two books changed how Suri thought about all of it. Eckhart Tolle’s The Power of Now taught him that he had three choices: change the situation, leave it, or accept it. Since he couldn’t change or leave his illness, he chose the hardest path — surrender.
Then, Anticancer by David Servan-Schreiber showed him how lifestyle could support healing. Suri overhauled his diet, embraced organic living, and attended a 40-day Vipassana meditation retreat. He has practiced daily meditation ever since.
“I stopped fighting to return to who I was,” he says. “I started focusing on being who I am now.”
Through back surgeries, heart surgery and multiple rounds of chemotherapy, including daily chemo medication for the last four years, Suri’s fear vanished. He completed an internship and passed his board exams to become a mental health counselor. It wasn’t just a career; it was a way to help humanity. It gave him a reason to stay.
Phase Four: “There Is Nothing Wrong with Me”
Today, Suri is 80 years old. He sees clients daily, teenagers, individuals, couples, and those in crisis, and even works pro bono. He occasionally writes poetry, exploring the idea that life and death are not opposites, but a “cosmic dance.”
Suri says that life and death exist in a continuous, harmonious cycle where millions of cells perish every second to make way for new growth. This constant process of cellular turnover ensures our survival, proving that our very existence depends on a delicate balance of ending and beginning. In a poem, Suri describes that balance:
No noise, no fear, just pure delight.
And life and death in harmony unite.
Where death appears not as a foe,
But as a friend where souls go.
“There is nothing wrong with me,” Suri says firmly. “Some people are born with six toes or crippling handicaps; they manage; I just have amyloidosis.”
He views the disease as a catalyst, not a curse. “I would never be this happy without it. I wouldn’t be the human I am.”
To those standing where he stood 20 years ago, Suri offers these words: “You didn’t do anything wrong. There is nothing you could have done differently. The only moment you can live is the one you are in. Live this moment fully.”
He Was Given 2 Years. He’s Giving 26.2 Miles.
In December 2006, Suri was given two years. This December, nearly 20 years later, he will walk the Honolulu Marathon to raise $15,000 for the Amyloidosis Research Consortium (ARC).
He is 80 years old and walking 26.2 miles. That is Phase Four.
Support Suri’s Journey: You can contribute to Suri’s marathon walk for research and patient support at arci.org/suri
Donate Today →
Need Support? The mental toll of amyloidosis is heavy. If you or a loved one are struggling with the emotional impact of a diagnosis, please call us at +1 (617) 467-5170 or email support@arci.org.









 Suri fighting at the hospital.
Suri fighting at the hospital.